Pharmacometrics & PK/PD modeling services
At Biotrial, we provide expert pharmacometrics consulting and quantitative clinical pharmacology services designed to support critical decision-making throughout drug development.
By integrating pharmacokinetic (PK) data, pharmacodynamic (PD) endpoints, and patient-level variability, modeling and simulation approaches allow sponsors to move beyond descriptive results and generate predictive insights that guide smarter development strategies.
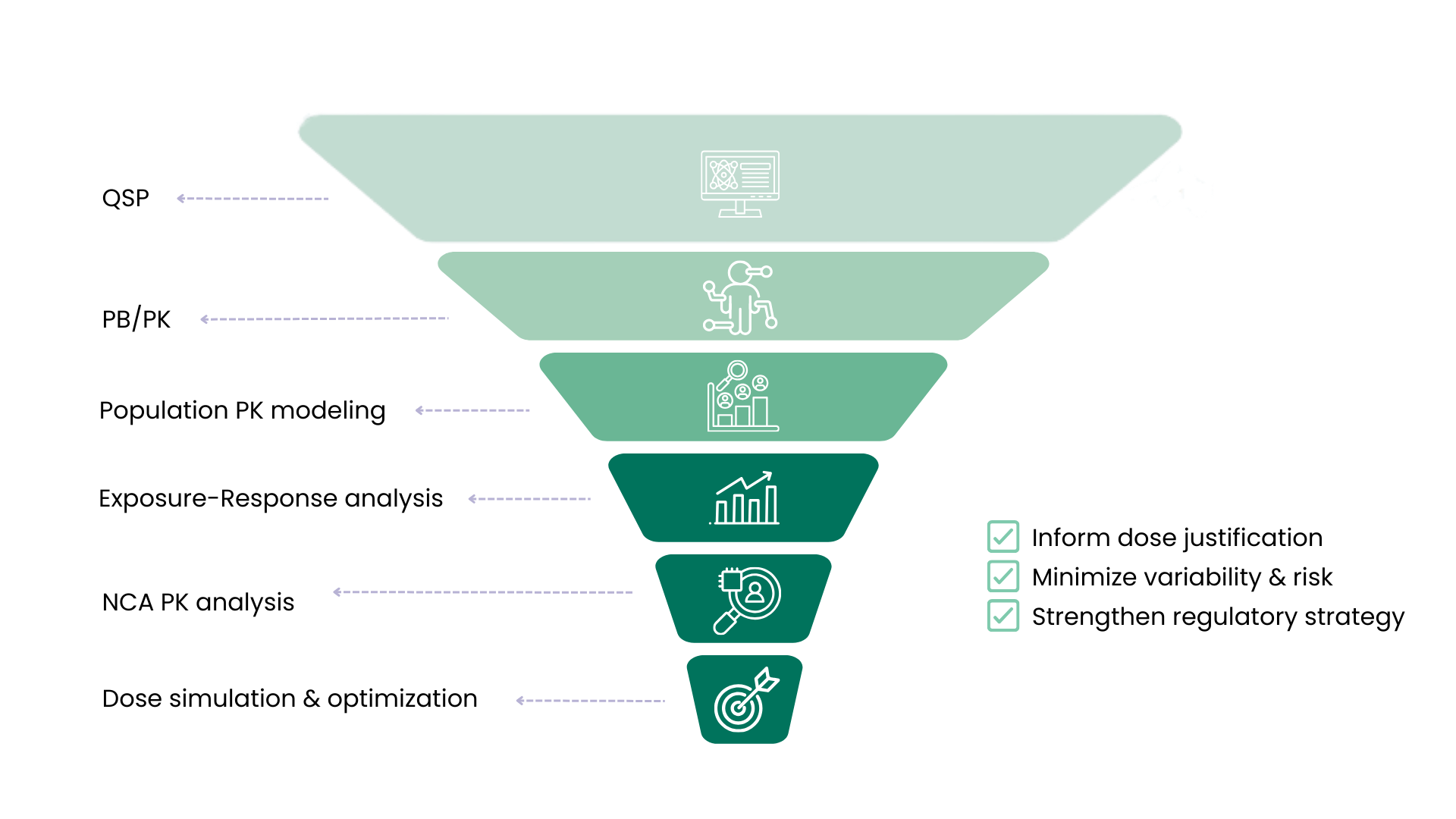
Our pharmacometricians translate complex datasets into actionable evidence through population PK (PopPK) modeling, exposure–response analyses and Physiological based pharmacokinetic modeling and simulation (PBPK). We incorporate these into Model-Informed Drug Development (MIDD) frameworks and clinical trial simulations. These approaches enable optimized dose selection, improved study design, reduced uncertainty, and stronger regulatory justification of development strategies.
Pharmacometrics services
Pharmacokinetic (PK) analysis
- Non-compartmental analysis (NCA) for robust summary PK metrics
- Compartmental PK modeling for mechanistic interpretation
- Interpretation of concentration–time data to support dose justification
- Deliverables formatted for regulatory submissions and review committees
Population PK/PK-PD modeling
- Population pharmacokinetics (PopPK) to quantify inter-individual variability
- Integrated PK-PD modeling linking exposure with efficacy and safety endpoints
- Exposure-response modeling to support risk/benefit optimization
- Clinical trial simulation to forecast outcomes under alternative dosing scenarios
Physiological based pharmacokinetic modeling and simulation (PBPK)
- Mechanistic models predicting human PK across populations, ages, organs, and disease states before first dosing.
- Integrating in vitro, preclinical, and clinical data for confident dose selection and DDI risk assessment.
- Simulating formulations, routes, food effects, organ impairment, and special populations.
- Regulatory-grade reporting aligned with FDA and EMA expectations, supporting waivers and labeling decisions.
Quantitative Systems Pharmacology (QSP)
- Mechanistic disease-drug models predictive of efficacy, biomarker response, and optimal patient populations.
- Integrating omics, literature, and clinical data to explain response variability.
- Simulate trials to explore combinations, dosing strategies, and enrichment criteria.
- Quantitative analyses support MIDD strategies, go/no-go decisions, and accelerated development pathways.
Model-Informed Drug Development (MIDD) & simulation
- Customized MIDD plans aligned with your compound’s development strategy
- PBPK and QSP modeling to support mechanistic understanding exposure in different (sub)populations
- Dose selection, escalation, and justification through simulation
- Simulation of virtual trials and scenario analyses to de-risk clinical decisions
Pharmacometric dose optimization
Regulatory-focused support
We prepare regulatory-ready quantitative packages compliant with global standards (FDA, EMA, PMDA) which will include the documentation for :
- Supporting the dose selection and labeling recommendations. All analyses and files required by regulatory agencies
- Submission-ready reports for INDs, NDAs, BLAs, and MAAs
- Clear, defendable quantitative evidence for regulatory discussions
Case study
Electroencephalography (EEG) recordings are extremely rich and can be complex to analyze. Modeling and simulation approaches for such data can bring additional key insights to help sponsors get the most out of these data.
One project our team worked on last year was a first in human trial collecting EEG data at many different dose levels. Such a study design provides very rich data for pharmacometricians to work with. In the case of this project, we were able to develop a PK/PD model investigating the qEEG response relative to the drug exposure and use this model to simulate responses or estimate sample size for future trials.

Applications across clinical development
Biotrial’s pharmacometric and PK/PD modeling expertise supports drug development across all clinical phases and therapeutic contexts. From preclinical and early First-in-Human (FIH) studies to late-stage registration programs, our quantitative approaches help sponsors optimize dose selection, anticipate variability, and strengthen evidence for key development decisions.
We routinely contribute to dose escalation and dose-ranging strategies, proof-of-concept trials, and exposure–response evaluations that inform labeling and benefit–risk discussions. Our team also supports specialized applications such as drug–drug interaction (DDI) assessments, pediatric development planning, and analyses in renal or hepatic impairment populations. When relevant, we can integrate real-world evidence or external data sources to complement clinical findings and enhance model-informed strategies.
Why to choose Biotrial
- Experienced pharmacometricians with strong clinical pharmacology expertise
- Integrated CRO environment ensuring seamless collaboration and data access
- Regulatory-aligned quantitative methodologies supporting FDA and EMA expectations
Tailored, decision-driven modeling strategies with clear, high-quality deliverables for teams and agencies

